“Their ability to understand complex topics and visualize them without face-to-face interaction was amazing.”
— Scott Stout, CEO & Co-Founder, MedVector
Clutch.co verified review
Between 2018 and 2021, Yum Yum Videos produced three custom explainer videos for MedVector, supporting a lean landing strategy built around video as the primary communication asset to present its telemedicine-based clinical trials model to investors and partners.
During this period, MedVector secured $3.5M in funding while relying on a focused landing page and video as its primary communication tools.

“Their ability to understand complex topics and visualize them without face-to-face interaction was amazing.”
— Scott Stout, CEO & Co-Founder, MedVector
Clutch.co verified review
Our collaboration with MedVector coincided with meaningful early-stage traction:
In this early-stage phase, Yum Yum Videos’ explainers served as a high-leverage communication asset—helping MedVector clearly explain a complex, regulated workflow through a lean, video-first approach.
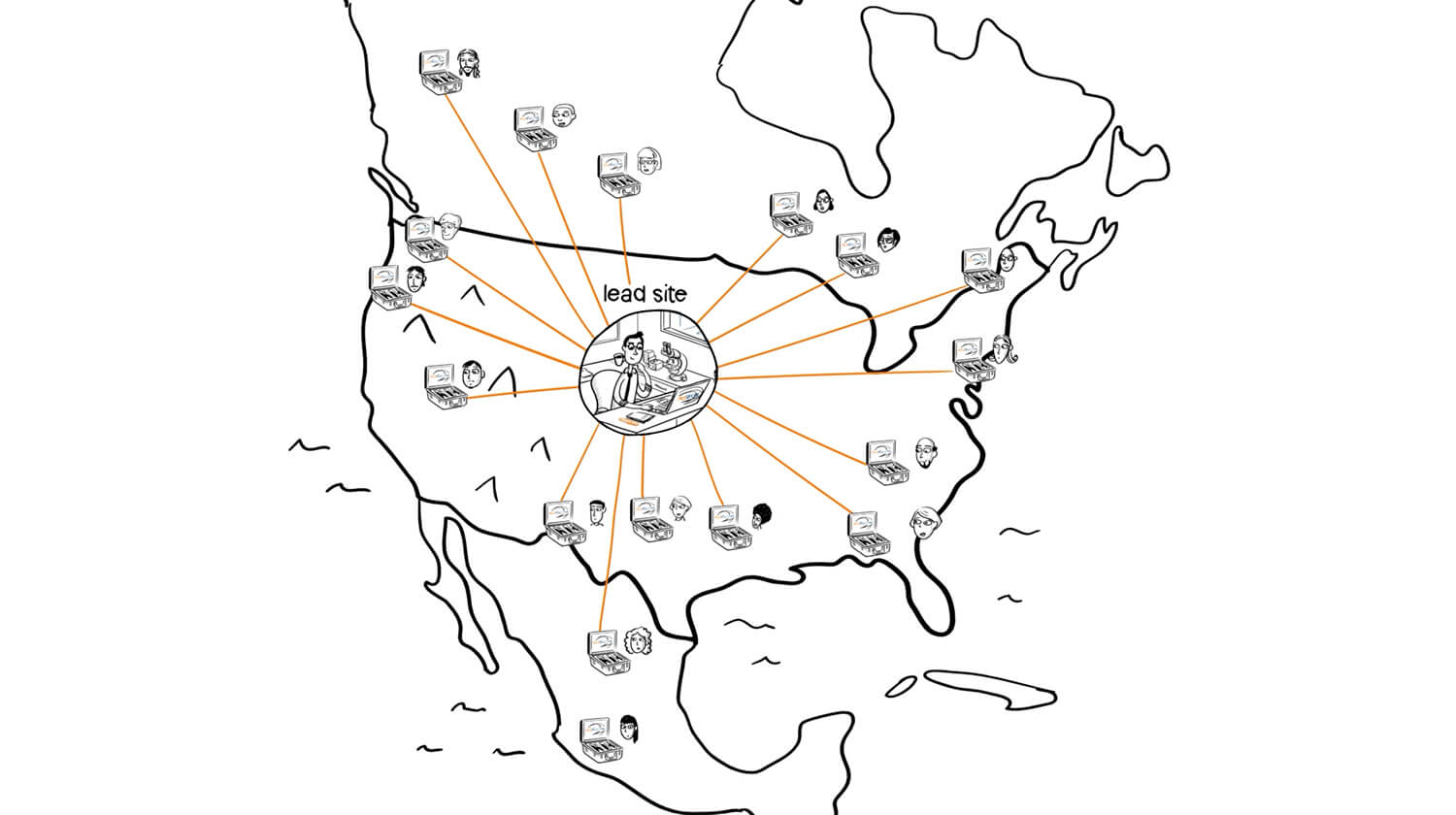
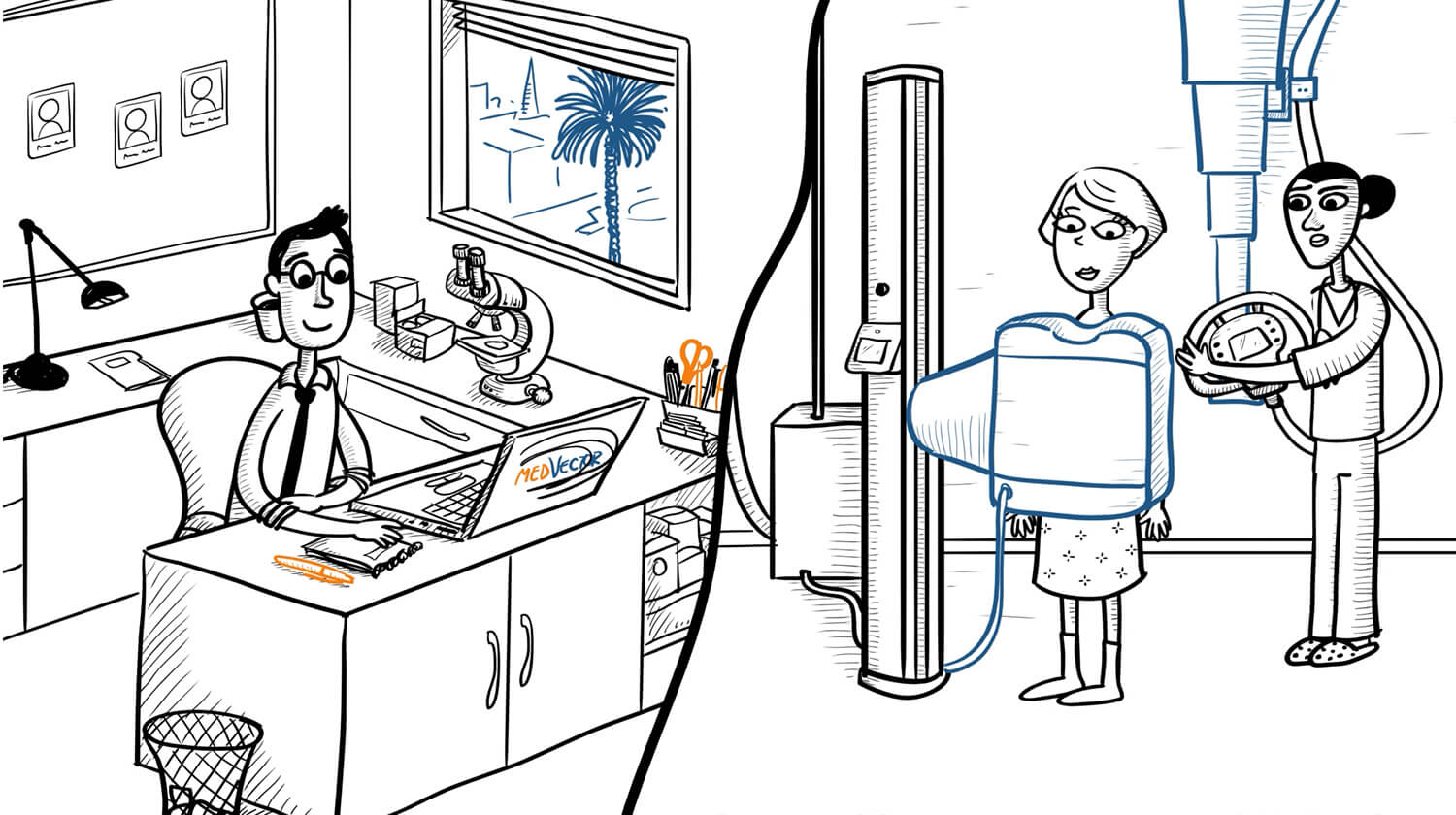
MedVector is a clinical trials enablement platform that allows eligible patients to complete study visits from their local physician’s office, while principal investigators conduct the visit remotely through HIPAA-compliant telemedicine.
By operating within existing clinical trial protocols, MedVector extends investigator reach and reduces patient travel burden — a differentiated but complex workflow requiring clear, precise communication to align investors, physicians, and institutional partners.
1. Explain a complex, regulated telemedicine workflow to multiple stakeholders.
2. Enable investor understanding using a lean, landing-page-driven strategy.
3. Align physicians, investigators, and partners around a new clinical-trials model.
To meet these needs, Yum Yum Videos created a focused series of three explainer videos between 2018 and 2021.
Each piece addressed a different layer of MedVector’s model—from introducing the core telemedicine workflow, to explaining physician participation and standardized data capture—allowing complex, regulated concepts to be communicated clearly through a single, video-centered landing page.
MedVector’s approach to decentralized clinical trials and telemedicine-enabled study visits was featured across investor platforms and healthcare-focused publications. These outlets highlighted how the company communicates a complex, regulated model to investors, physicians, and partners—often using video as a core explanatory asset.
“MedVector offering page featuring the explainer video as the primary communication tool.”
“MedVector to Connect Patients of Local Physicians to Study Investigators”
“MedVector to Clarify Misunderstood FDA Guidance at J.P. Morgan Healthcare Conferences”
“Product page using the explainer video to present MedVector’s clinical trials workflow.”
From strategy to execution, every detail was designed to turn a complex, regulated model into a clear and credible story.
1. Clarity under regulation – Breaking down a multi-stakeholder, HIPAA-compliant workflow into simple, visual narratives.
2. Consistency across assets – Three explainer videos, all aligned under one clear message and visual language.
3. High leverage – A single set of videos powering a focused landing page, investor outreach, and partner conversations.
4. Investor-ready storytelling – Videos designed to support understanding during fundraising and early-stage growth.
A clear whiteboard narrative transformed a complex, regulated clinical-trials workflow into an easy-to-follow visual story.
Clean line illustrations, purposeful motion, and minimal visual elements kept the focus on process and understanding rather than decoration.
A restrained whiteboard palette—black and gray linework with selective accent color—reinforced clarity while maintaining a clinical, professional feel.
Calm, trustworthy, and precise, supporting credibility with investors, physicians, and institutional partners.